Insights & Best Practices for Pharma Professionals
Stay up to date with the latest developments, trends, and best practices in the pharmaceutical industry.
Pharmaceutical Import Germany: Three Legal Pathways
Pharmaceutical import into Germany is governed by a precise regulatory framework that creates both obligations and opportunities for international traders. Whether you are pursuing parallel import, individual import under Article 73.3 AMG, or exploring niche products...
Why Germany Is the Gateway Market for European Pharma Access
German pharma market access data is the foundation of every pricing, reimbursement, and launch decision for pharmaceutical companies entering Europe's largest single-country drug market. Germany generates over EUR 60 billion in annual pharmaceutical revenue at...
Why Pharmaceutical Data APIs Are Becoming Essential
A pharmaceutical data API is a programmatic interface that delivers structured drug information -- PZN identifiers, pricing, active ingredients, dosage forms, and clinical data -- directly into your software. In 2026, with Germany's electronic patient record (ePA)...
What Is an EU Drug Authorization Database?
An eu drug authorization database is a consolidated platform that lets pharmaceutical professionals search marketing authorizations across multiple countries from a single interface. Instead of navigating dozens of national registries in different languages, you query...
German Drug Pricing Data: What International Teams Need to Know
German drug pricing data is among the most requested datasets in global market access. Germany is the largest pharmaceutical market in Europe, the fourth-largest worldwide, and serves as a reference price country for around 16 European countries. With statutory health...
How Drug Shortages Are Reported in Germany
A drug shortage database for Germany is built on a legally mandated reporting framework that determines what data is available, how current it is, and where the gaps are. Understanding this framework is essential for any pharma professional who wants to turn shortage...
Why Pharmaceutical Data APIs Are Becoming Essential
A pharmaceutical data API is the most efficient way to integrate structured drug information — PZN identifiers, pricing, active ingredients, and clinical data — into your own software. Whether you are building a health-tech application, an ERP module, or a clinical...
What Is an EU Drug Authorization Database?
An EU drug authorization database is the single most important research tool for pharmaceutical professionals who need to know which medicines are authorized in which countries. Whether you are sourcing reference products for a generic application, identifying market...
How Drug Pricing Works in Germany
German drug pricing data follows a regulated, multi-stage calculation chain that determines what manufacturers, wholesalers, pharmacies, and patients pay for prescription medicines. Unlike market-driven pricing systems in the United States, Germany uses a...
Why Discontinued Drugs Matter for Pharma Business Development
A discontinued drugs Germany archive is one of the most overlooked resources in pharmaceutical business development. Every year, dozens of medicinal products are deleted from the German market -- and as of March 2026, BfArM lists 562 active shortage entries, many of...
Navigating the Transatlantic Pharmaceutical Landscape: A Comprehensive Analysis of ABDA Standards, FDA Mutual Recognition, and Market Access in Germany (2025)
Executive Summary: The Dual Challenge of Compliance and Strategy The global pharmaceutical landscape of 2025 is defined by a paradoxical dynamic: regulatory harmonization on one hand, and increasing market complexity on the other. For pharmaceutical professionals...
The Central Nervous System of Modern Healthcare: A Comprehensive Analysis of Medication Databases, Market Access, and Clinical Safety Infrastructure
Executive Summary In the contemporary healthcare ecosystem, the medication database has transcended its traditional role as a static repository of pharmaceutical nomenclature. It has evolved into a dynamic, mission-critical infrastructure that underpins patient...
The Iceberg Search: Navigating the Submerged Depths of Pharmaceutical Intelligence and Drug Databases
1. The Epistemology of Drug Data: Beyond the Visible Web In the pharmaceutical and healthcare industries, information is not merely a utility; it is the fundamental currency of survival, compliance, and therapeutic success. When a regulatory affairs manager, a...
The Strategic Imperative of the Summary of Product Characteristics (SmPC): Regulatory Frameworks, Digital Transformation, and AI-Driven Compliance
Executive Summary The global pharmaceutical ecosystem is currently navigating a period of unprecedented complexity. At the epicenter of this environment—balancing the rigorous demands of regulatory compliance, the clinical needs of healthcare professionals, and the...
Find German Equivalent of US Drugs – A Guide for Pharma Professionals
Find German equivalent of US drugs – it’s a challenge that pharmacists, healthcare providers, and pharmaceutical analysts often face. A patient arrives with a U.S.-branded prescription, or a formulary review requires checking if an American medication is available in...
AMNOG Reimbursement Database Europe – Unlocking Pricing & Access Data for DACH Markets
In the pharmaceutical industry, having timely and accurate reimbursement data is crucial for successful market access across Europe. Especially in Germany, Austria, and Switzerland (DACH), you face a patchwork of pricing systems and databases. Navigating these can be...
Drug Shortage Database Germany – A Comprehensive Solution to Medicine Supply Gaps
Germany has been grappling with increasing medicine shortages in recent years. Ensuring a reliable drug shortage database in Germany is crucial for healthcare stakeholders to maintain patient care and plan effectively. Official figures show that medicine supply issues...
OTC and Rx Drug Database Germany: The Complete Guide for Pharma Professionals
Entering Germany’s pharmaceutical market requires navigating a vast landscape of prescription (Rx) and over-the-counter (OTC) drug information. From regulatory nuances to pricing and reimbursement details, having all data at your fingertips is crucial. An OTC and Rx...
Pharmacy Wholesale Drug Prices in Germany: Ensuring Transparency, Access, and Compliance
Pharmacy wholesale drug prices in Germany are a critical concern for international pharmaceutical wholesalers looking to stay competitive and compliant. Germany is Europe’s largest pharmaceutical market, and its drug pricing system is highly regulated – making market...
Drug Pricing Database Germany: A One-Stop Solution for Up-to-Date Pharmaceutical Prices
Drug pricing database Germany – these four words sum up a critical need in the pharmaceutical industry. In a market as regulated and dynamic as Germany’s, professionals across market access, pharmacovigilance, hospital pharmacy, and health insurance face the constant...
Pharma Pricing Germany: Navigating Costs, Compliance and Transparency
Pharmaceutical pricing in Germany is unique in Europe – offering initial free pricing yet governed by strict mechanisms like AMNOG benefit assessments, reference pricing, and rebate contracts. In this comprehensive guide, we explain pharma pricing in Germany for...
European Drug Pricing Database: Comprehensive Pricing Insights for Healthcare Professionals
Access to an accurate, up-to-date European drug pricing database is essential for healthcare professionals in Germany, Austria, and Switzerland. Whether you’re calculating reimbursement, comparing market prices, or sourcing alternatives during shortages, having...
Pharmaceutical Market Access Germany: Navigating Approval, Pricing, and Reimbursement
Pharmaceutical market access in Germany is a critical step for any drug developer aiming to tap into Europe’s largest pharmaceutical market. With a population of 84 million and annual pharmaceutical revenues exceeding €56.9 billion in 2021, Germany offers vast...
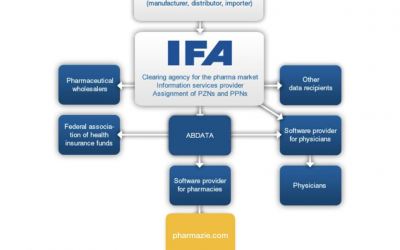
IFA Pharmaceuticals and their way into pharmaceutical databases
IFA Pharmaceuticals and their way into pharmaceutical databasesHow does the IFA medicines information get from the ABDA article master to pharmaceutical databases like pharmazie.com? This question is answered in the following article. You will learn who the IFA is,...
The Planetary Health Diet
The Planetary Health Diet 37 scientists from 16 countries have gazed into the future, and it will not work without our help! You can read in this article how exactly this diet and the rescue of the planet will work. Today we are confronted with an ever-increasing...
The new packaging law 2019 – who is affected?
The new Falsified Medicines Directive 2019 – which medicines are affected?
The new Falsified Medicines Directive 2019 - which medicines are affected? As of 9. February 2019, the Falsified Medicines Directive (2011/62/EU), which was adopted in 2011, has officially come into force. What does this mean for the affected players such as...
Estimating the final price of a prescription-only product
Estimating the final price of a prescription-only product At a glance: Pricing for pharmaceuticals in Germany Supply and demand determine the price, at least considering most branches of a free market economy. This basic principle also applies to non-prescription...
ABDA-Database
Marketing Authorisation Holder Drug Dictionaries (ABDA-Database)The ABDA-database is a facility device for pharmaceutical and medical experts for comprehensive information on original pharmaceutical products and generics available in Germany, Switzerland, Austria and...
Drug Pricing Germany Database ABDA Artikelstamm Taxe
Drug Pricing Tool German Medicinal Products - ABDA-Artikelstamm (Taxe)The Drug Pricing Tool German Medicinal Products - ABDA-Artikelstamm (Taxe) includes all economic data for all drugs sold in pharmacies in Germany. Pressed to make pricing decisions? Need to...
Our Databases – Overview
Our Databases: OverviewSince 1989 [pharmazie.com] offers comprehensive information on medication for medical and pharmaceutical experts in at the time 35 databases. Some of these databases are free of a license and costs, others require a license. The tool...